Three Acid-Base Theories
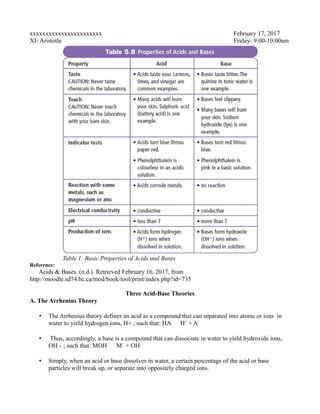
- 1. xxxxxxxxxxxxxxxxxxxxxxx February 17, 2017 XI- Aristotle Friday- 9:00-10:00am Reference: Acids & Bases. (n.d.). Retrieved February 16, 2017, from http://moodle.sd74.bc.ca/mod/book/tool/print/index.php?id=735 Three Acid-Base Theories A. The Arrhenius Theory • The Arrhenius theory defines an acid as a compound that can separated into atoms or ions in water to yield hydrogen ions, H+ ; such that: HA H+ + A- • Thus, accordingly, a base is a compound that can dissociate in water to yield hydroxide ions, OH - ; such that: MOH M+ + OH- • Simply, when an acid or base dissolves in water, a certain percentage of the acid or base particles will break up, or separate into oppositely charged ions. Table 1: Basic Properties of Acids and Bases
- 2. • Furthermore, acids and bases will neutralize each other when mixed. They produce water and an ionic salt, neither of which are acidic or basic; such that: HA + MOH H2 0 + MA B. The Brönsted-Lowry Theory • The theory provides a more general definition of acids and bases that can be used to deal both with solutions that contain no water and solutions that contain water. • An acid is a substance from which a proton (H+ ion) can be removed. • A base is a substance to which a proton (H+) can be added. • Essentially, it defines an acid as a proton donor and a base as a proton acceptor. ◦ “When an acid loses a proton, the remaining species can be a proton acceptor and is called the conjugate base of the acid. Similarly when a base accepts a proton, the resulting species can be a proton donor and is called the conjugate acid of that base.” • On the other hand, water, H2O, can be considered an acid or a base since it can lose a proton to form a hydroxide ion, OH - , or accept a proton to form a hydronium ion, H3O+. C. The Lewis Theory • Accordingly, the Lewis definition is the most general theory, having no requirements for solubility or protons. • In this theory, an acid as a compound that can accept a pair of electrons and a base as a compound that can donate a pair of electrons. ◦ “Lewis acids and bases react to create an adduct, a compound in which the acid and base have bonded by sharing the electron pair.” References: Acid-Base Theories. (2012). Retrieved February 16, 2017, from http://www.infoplease.com/encyclopedia/science/acids-bases-acid-base-theories.html Brown, Theodore L. ... et al, Brown, T. L., LeMay, J. H., Bursten, B. E., Murphy, C. J., & Woodward, P. -. (2009). Chemistry: the central science. - 11th Edition. Upper Saddle River, NJ: Pearson Prentice Hall.
